How to THRIVE in the Operating Theatre

It could be argued, in recent years few other changes in practice or adoptions of new techniques have the potential to positively impact patient safety and outcomes in the operating theatre, more than the THRIVE technique. This novel use of an established ‘ward based’ respiratory therapy, High Flow Oxygen Therapy (HFOT), applied to the theatre setting has gained a foothold across numerous hospitals in European countries, and beyond, with many now using it as standard practice for preoxygenation and safer intubation. In this blog we will bring it back to basics. What is the THRIVE technique? How does it work? Why would you consider using it?
Let's break it down...
The acronym is always a good place to start. THRIVE stands for Transnasal Humidified Rapid-Insufflation Ventilatory Exchange and was first coined in the UK by Patel and Nouraei during a study conducted by the pair between 2013 and 2014 [1]. As the name suggests, the technique is delivered to a patient via the nasal passages (Transnasal) using specialist equipment. It utilises oxygen rich gases, that have been heated and had water vapour added to ‘optimally condition’ them (Humidified), blown into the upper airways at flow rates in excess of 45L/pm (Rapid-Insufflation). These humidified gases are then used to oxygenate a patient during apnoea following induction of anaesthesia (Ventilatory Exchange).
Originally used in the acute care ward setting, high flow rates of respiratory gas, at a prescribed and set FiO2, have been shown to aid in the treatment of patients with hypoxaemia. The easy to fit and operate equipment [Image 1 & 2] has made strides in the treatment of patients with Type 1 respiratory failure, bridging the gap between low flow oxygen and CPAP therapies and is widely referred to as High Flow Oxygen Therapy (HFOT) [2]. The development of the THRIVE technique, using HFOT, has therefore paved the way for the diversification of this therapy into alternative hospital departments. In the theatre setting it not only enables the use of the THRIVE technique but also demonstrates additional benefits that may be useful throughout the perioperative journey.

So, what does HFOT do that makes it particularly useful in the theatre setting?
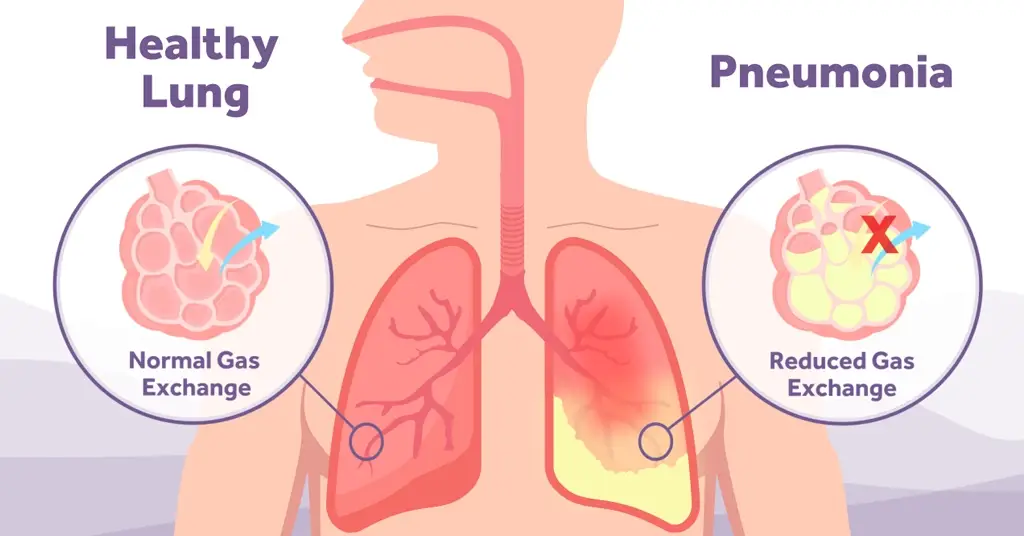
HFOT works by washing out the upper airways and generating a humidified fresh gas reservoir that leads to multiple benefits for the patient. This ‘nasopharyngeal dead space washout’ is achieved by the high flows of gas being delivered directly into the upper airway from the purpose-built high flow nasal cannula [3]. The oxygen rich gas displaces waste gases normally found in the nasopharyngeal area and replaces them with constantly refreshed gases delivered at the set FiO2. Benefits to patient include improved alveolar ventilation, accurate FiO2 delivery, reduced inspiratory work of breathing, variable PEEP effect, improved mucociliary clearance and patient comfort and the ability to deliver apnoeic oxygenation [4,5].
In turn, each of these benefits may have a positive impact directly in the operating theatre environment. The improvement in alveolar ventilation, because of the reduced anatomical dead space in the upper airways, helps contribute to better preoxygenation due to the increased volume of O2 available in the alveoli for gaseous exchange [3]. Accurate FiO2 delivery, through less dilution of inspired gases due to reduced CO2 rebreathing from the anatomical dead space and diminished entrainment of room air, creates a greater concentration of O2 in the alveoli contributing to better preoxygenation [3].
Reduced inspiratory work of breathing is useful in post-op recovery to help ensure adequate oxygenation and recovery in a situation where respiratory effort may not be optimal. The variable PEEP effect, generated by the patient’s expiratory breath having to pass through partially occluded nares against oncoming gas flow [6], may also be useful in post-op recovery to recruit alveoli in patients suffering from post-op atelectasis and during preoxygenation in patients who have existing atelectasis, ensuring full lung patency for enhanced gaseous exchange. The heating and addition of water vapour to the respiratory gases used in HFOT helps improve mucociliary clearance and patient comfort [7].
The theatre environment being normally colder than ward can impact normothermia. Having a heated therapy, especially in recovery, can help the patient return to normothermia more quickly. The ability of HFOT to deliver apnoeic oxygenation is where the THRIVE technique comes into play. By utilising a physiological phenomenon known as Aventilatory Mass Flow (AVMF), HFOT can be used in theatre to extend the time a patient remains within normal oxygen saturation range while apnoeic [1].
Aventilatory Mass Flow (AVMF)
This Aventilatory Mass Flow (AVMF) [8] phenomenon, present during the THRIVE technique, allows for extended apnoea time within a safe oxygen saturation range. The fresh gas reservoir created by HFOT ensures constant availability of oxygen in the upper airways that is drawn into the lung due to the negative pressure gradient that develops in the lower airways during apnoea [1,9]. With the rate of O2 removal from the alveoli into the blood capillaries unchanged following induction of apnoea, the reduced CO2 excretion that occurs results in a generation of the negative pressure due to lack of ventilatory movement [8,9]. As long as the patient’s airway remains open, a pathway exists to atmosphere and the pressure gradient draws O2 from the fresh gas reservoir [10].
Conclusion
Using the THRIVE technique and AVMF allows for continual oxygenation of a patient for lengths of time far exceeding the standard apnoeic window. It has been shown to extend safe apnoea time up to 65 minutes [1] opening the door to tubeless operations, especially beneficial in ENT surgery and biopsies. The benefits to the theatre department in terms of increased patient safety and throughput of cases are just some of the positive impacts the introduction of HFOT to deliver THRIVE can present. Further exploration for individual needs may be required but certainly even the benefit of improved and effective preoxygenation will transpire across the hospital with additional benefits applying in most cases.